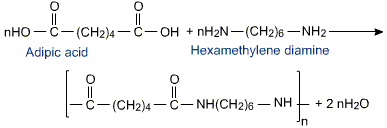Preparation of Nylon 66
The polyamide Nylon 66 is formed by heating the reactant mixture under pressure and the process has been developed so that the molecular mass of the polymer is controlled in the range of 12,000 to 20,000 amu.
Nylon-6 is prepared from the monomer caprolactam which is obtained from cyclohexane (petrochemical). Since caprolactam is more easily available, it is used for polymerization which is carried out in the presence of water that first hydrolyzes the caprolactam to the amino acid. Subsequently, the amino group of the amino acid can react with caprolactam to form the polyamide polymer.
Filaments of Nylon-6 are obtained by melt-spinning of the polymer. The fibers are cooled by a stream of air.
Uses of nylon-:
Historical uses
- Bill Pittendreigh, DuPont, and other individuals and corporations worked diligently during the first few months of World War II to find a way to replace Asian silk and hemp with nylon in parachutes.
- It is also used to make tires, tents, ropes, ponchos, and other military supplies.
- Nylon can be used as the matrix material in composite materials, with reinforcing fibers like glass or carbon fiber; such a composite has a higher density than pure nylon. Such thermoplastic composites (25% to 30% glass fiber) are frequently used in car components next to the engine, such as intake manifolds, where the good heat resistance of such materials makes them feasible competitors to metals.
- It is used as elastomers
- It is used as plastics
- It is used in toys.
Various nylons break down in fire and form hazardous smoke, and toxic fumes or ash, typically containing hydrogen cyanide. Incinerating nylons to recover the high energy used to create them is usually expensive, so most nylons reach the garbage dumps, decaying very slowly. Some recycling is done on nylon, usually creating pellets for reuse in the industry.
Preparation of Nylon 66 and its Practical Procedure:
Related Links – Uses of Nylon 66 and Properties of Nylon 66