Properties of Nylon 66
- Nylon 66 has a repeat unit with a molecular weight of is 226.32 g/mol and crystalline density of 1.24 g/(cm)^3 .
- Nylon 66 has long molecular chains resulting in more hydrogen bonds, creating chemical springs and making it very resilient.
- Nylon 66 is an amorphous solid so it has a large elastic property and is slightly soluble in boiling water.
- Nylon 66 is very stable in nature.
- Nylon 66 is very difficult to dye but once it is dyed it has a high colorfastness and is less susceptible to fading.
- Its chemical properties do not allow it to be affected by solvents such as water, alcohol etc.
- Glass transition temperature: 50oC.
- Chemical formula: (C12H22N2O2)n
-
Density: 1.314 g/mL (Zytel)
- Melting point: 507 °F (264 °C)
- Melting temperature: 255oC.
- Amorphous density at 25oC: 1.07 g/cm3.
- Crystalline density at 25oC: 1.24 g/cm3.
- The molecular weight of repeat unit: 226.32 g/mol.
Synthesis and manufacturing of Nylon 66:
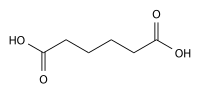 |
Nylon 66 is synthesized by polycondensation of hexamethylenediamine and adipic acid. Equivalent amounts of hexamethylenediamine and adipic acid are combined with water in a reactor. This is crystallized to make nylon salt, which has precisely stoichiometric equivalents. The nylon salt goes into a reaction vessel where polymerization process takes place either in batches or continuously. Removing water drives the reaction
n HOOC-(CH2)4-COOH + n H2N-(CH2)6-NH2 → [-OC-( CH2)4-CO-NH-(CH2)6-NH-]n + 2n H2O
toward polymerization through the formation of amide bonds from the acid and amine functions. Thus molten nylon 66 is formed. It can either be extruded and granulated at this point or directly spun into fibers by extrusion through a spinneret (a small metal plate with fine holes) and cooling to form filaments.
Properties of Nylon 66:
Related Links: Preparation of Nylon 66 and Nylon 66 Uses

