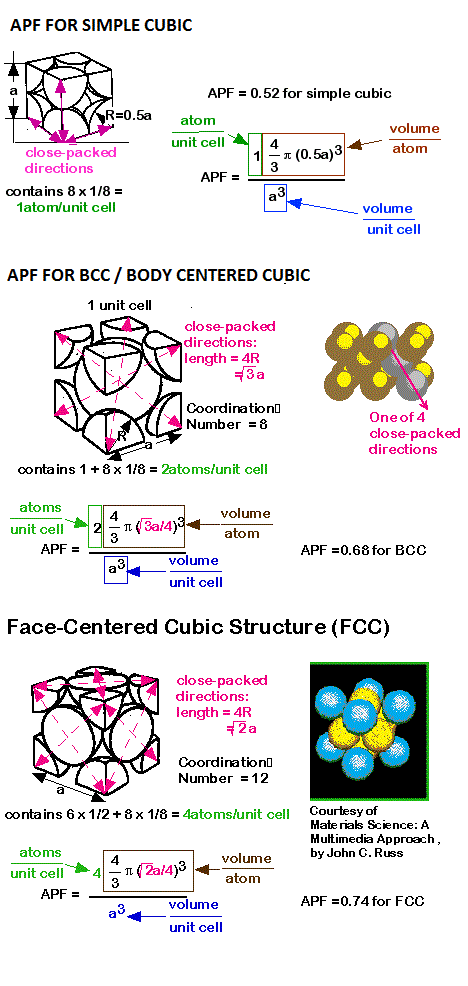
Atomic Packing factor for SC BCC FCC and HCP
In crystallography, atomic packing factor (APF), packing efficiency or packing fraction is the fraction of volume in a crystal structure that is occupied by constituent particles. It is dimensionless and always less than unity. In atomic systems, by convention, the APF is determined by assuming that atoms are rigid spheres. The radius of the spheres is taken to be the maximal value such that the atoms do not overlap. For one-component crystals (those that contain only one type of particle), the packing fraction is represented mathematically by
 { atomic packing factor for sc bcc fcc }
{ atomic packing factor for sc bcc fcc }
where Nparticle is the number of particles in the unit cell, Vparticle is the volume of each particle, and Vunit cell is the volume occupied by the unit cell.
It can be proven mathematically that for one-component structures, the most dense arrangement of atoms has an APF of about 0.74, obtained by the close-packed structures. For multiple-component structures, the APF can exceed 0.74.
The expression to calculate atomic packing factor is = (No. of atoms/cell) x (volume of each atom) divided by (volume of an atom)
People Also Searched for:
Conservation of Natural Resources
Already we know that natural resources are exhausting rapidly, we must conserve for future generation. so its duty of individual to conserve natural resources.
Sabine Formula
Sabine’s reverberation equation was developed in the late 1890s in an empirical fashion. He established a relationship between the RT60 of a room, its volume, and its total absorption (in sabins).
Depression of a Cantilever
A cantilever is a rigid structural element, such as a beam or a plate, anchored at only one end to a (usually vertical) support from which it is protruding.
Atomic Packing factor
In crystallography, atomic packing factor (APF), packing efficiency or packing fraction is the fraction of volume in a crystal structure that is occupied by constituent particles.
You can share your feedback (both positive and negative) by mailing to [email protected]. It will help us to improve our site.We will respond your mail within 24 hours.