Free Energy
Gibbs Free Energy (G) – The energy associated with a chemical reaction that can be used to do work. The free energy of a system is the sum of its enthalpy (H) plus the produ犀利士5mg
ct of the temperature (Kelvin) and the entropy (S) of the system.
Free energy of reaction (G)
The change in the enthalpy (H) of the system minus the product of the temperature (Kelvin) and the change in the entropy (S) of the system
Standard-state free energy of reaction (G)
- The free energy of reaction at standard state conditions:
Standard-state conditions
- The partial pressures of any gases involved in the reaction is 0.1 MPa.
- The concentrations of all aqueous solutions are 1 M.
Tabulated standard-state thermodynamic data are generally for a temperature of 25C (298 K)
Standard-State Free Energy of Formation (Gf)
- The change in free energy that occurs when a compound is formed from its elements in their most thermodynamically stable states at standard-state conditions. In other words, it is the difference between the free energy of a substance and the free energies of its elements in their most thermodynamically stable states at standard-state conditions.
- The standard-state free energy of reaction can be calculated from the standard-state free energies of formation as well. It is the sum of the free energies of formation of the products minus the sum of the free energies of formation of the reactants:
Recall from the enthalpy notes that reactions can be classified according to the change in enthalpy (heat):
Endothermic – absorbs heat, H > 0
Exothermic – releases heat,H < 0
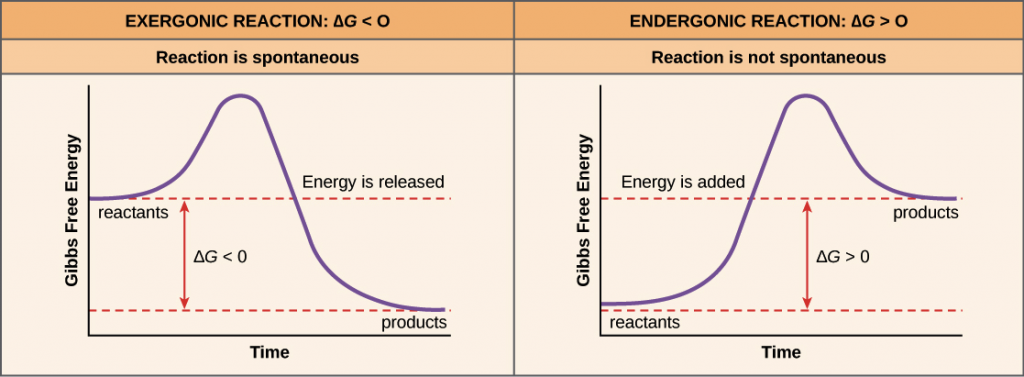
Reactions can also be classified according to the change in the free energy of the reaction:
Endergonic – NON-SPONTANEOUS, G > 0
Exergonic – SPONTANEOUS, G < 0